Microfluidic Hydrodynamic Voltammetric Techniques
Within the group we typically use a number of voltammetric techniques (both DC and AC), the fundamentals of which are explained in the teaching notes. Hydrodynamic devices have been employed for an enormous variety of electrochemical applications in recent years, including mechanistic investigations and analytical detection. In the field of electrochemistry tools such as the rotating disc, channel and wall-jet electrodes have provided key insights into properties such as the composition, chemical and photochemical reactivity of molecules in fluid solution. However despite their undoubted success these hydrodynamic tools share a number of common limitations. One of the most significant of these is the inability to mix reagents in a well defined way close to the detection region. In many cases the various reactants are held within the same vessel which severely limits the application of these techniques to quantitative mechanistic and kinetic investigation of rapid chemical reactions. We have been involved in the development of a novel device, the confluence reactor, which overcomes some of the traditional limitations associated with hydrodynamic devices. The confluence reactor consists of two rectangular inlets supplied with reactants from different reservoirs as shown in the diagram below.
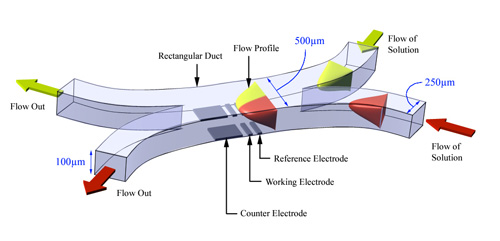
The microfluidic confluence reactors are produced in house using standard microfabrication procedures. They are designed to allow the fluid to reach a well defined laminar and parabolic flow profile prior to mixing. By using relatively simple microfabrication techniques it is easy to alter both the size and number of the inlets for use with different systems. Simulation of the convective and diffusive properties using finite element techniques has also enabled the current response of chemically unreactive species from the separate inlets to be modelled as a function of the mass transport rate. The confluence reactor has also been used in conjunction with imaging techniques in order to provide experimental understanding of the concentration distributions arising from hydrodynamic voltammetric measurements.

